

1. The Dual Corrosion Protection Mechanism of Zinc Coatings
The protection provided by a galvanized zinc coating to steel relies primarily on a physical barrier and electrochemical protection. The physical barrier serves as the most direct form of protection for the steel; the zinc layer completely isolates the steel substrate from corrosive media such as air, moisture, and salts, thereby preventing contact between the steel and the corrosive environment at the source. The corrosion products of zinc (ZnO/Zn(OH)₂/ZnCO₃) further form a dense passivation film, enhancing the isolation effect.
Electrochemical protection is the most critical aspect of galvanized protection. The electrode potential of zinc is -0.76 V, while that of iron is -0.44 V. Zinc is more chemically reactive than iron. When the galvanized coating suffers minor damage and the steel substrate is exposed to corrosive agents such as rainwater or moisture, zinc acts as the “anode” and is corroded first, while the exposed steel acts as the “cathode” and is protected. In essence, zinc sacrifices itself to ensure the safety of the steel.
The service life of the protective coating is strongly correlated with the thickness of the zinc layer: since the corrosion rate of zinc is relatively stable (approximately 5–10 μm per year in natural outdoor environments), a thicker zinc layer results in a longer service life for the protective barrier. Industry data indicates that each micrometer of zinc coating provides 1–2 years of effective protection.
This process is known as cathodic protection or sacrificial anode protection. Even if the zinc coating wears away in certain areas, the surrounding zinc continues to protect the exposed iron through electrochemical action until the zinc layer is completely depleted. This is precisely why the cut edges of galvanized products are resistant to rust.
2. Comparison of Mainstream Galvanizing Processes:Hot-Dip Galvanizing vs. Electrogalvanizing
2.1 Hot-dip galvanizing
Hot-dip galvanizing (also known as hot-dip zinc coating) is a galvanizing process in which the surface of cleaned steel is immersed in molten zinc, causing a chemical reaction between the zinc and the steel surface to form a zinc-iron alloy layer. The advantages of hot-dip galvanizing include a thick coating, excellent corrosion resistance, and high mechanical properties. It is suitable for manufacturing heavy-duty components of precision castings and parts with high corrosion resistance requirements, and is widely used in the construction, bridge, automotive, and marine industries.
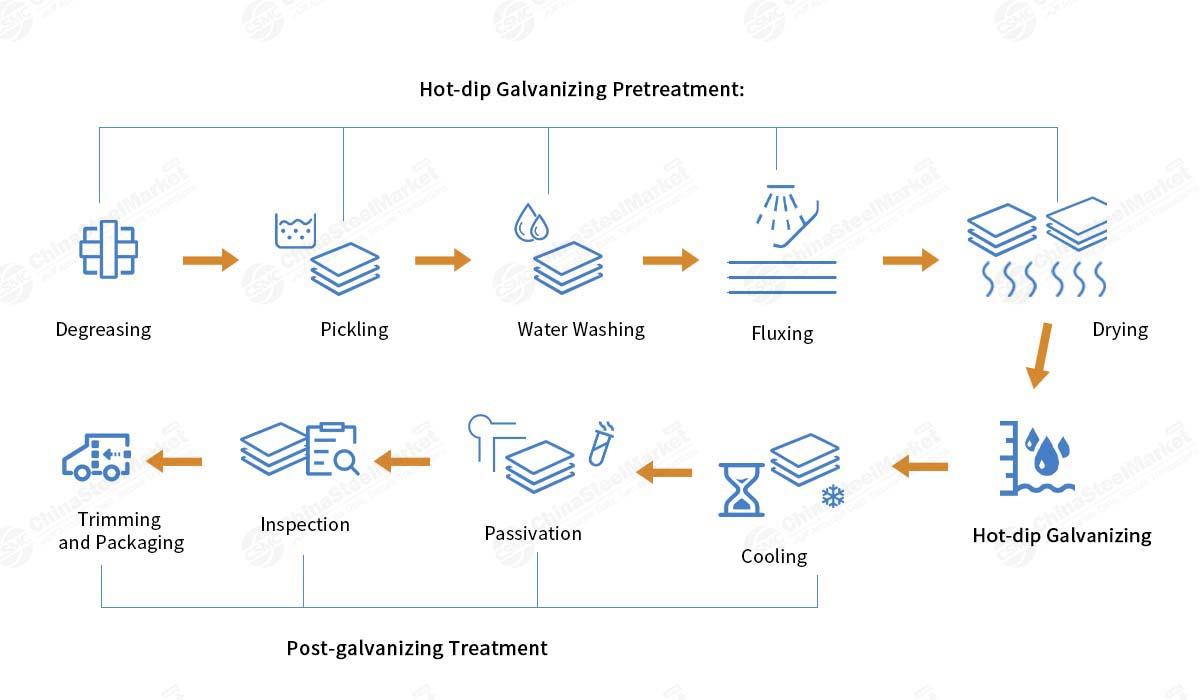
The following are the detailed process steps.
Surface pretreatment: Includes degreasing, pickling, rinsing, and fluxing.
Degreasing: Removes oil and impurities from the steel surface.
Pickling: Removes oxides and rust layers from the steel surface.
Fluxing: Forms a pre-plating film on the steel surface to improve the adhesion of the zinc coating during galvanizing.
Hot-dip galvanizing: The pretreated steel is immersed in molten zinc at 450–480°C. The steel reacts chemically with the zinc to form an alloy layer, which is ultimately covered by a layer of pure zinc.
Cooling and post-treatment: After hot-dip galvanizing, the steel undergoes cooling, passivation, and any necessary post-treatment to improve its corrosion resistance and surface finish.
2.2 Electro-Galvanizing
Electro-galvanizing (also known as cold galvanizing) is a process that uses electrochemical deposition to form a zinc coating on the surface of steel components. Unlike the physicochemical reaction involved in hot-dip galvanizing (immersion in molten zinc), it can form a uniform, dense zinc layer with excellent corrosion resistance on the surface of precision castings. Electro-galvanizing involves using an external current to reduce zinc ions in an electrolyte containing zinc ions and deposit them onto the steel surface acting as the cathode, resulting in a pure zinc layer (typically without an alloying layer). The advantages of electrogalvanizing include uniform coating thickness and an attractive, lustrous finish. It is suitable for surface protection of precision castings and is commonly used in the manufacture of furniture, appliances, and automotive components.
The coating is relatively thin (typically 5–30 micrometers), so its corrosion resistance is somewhat limited; however, it offers excellent decorative properties and workability.
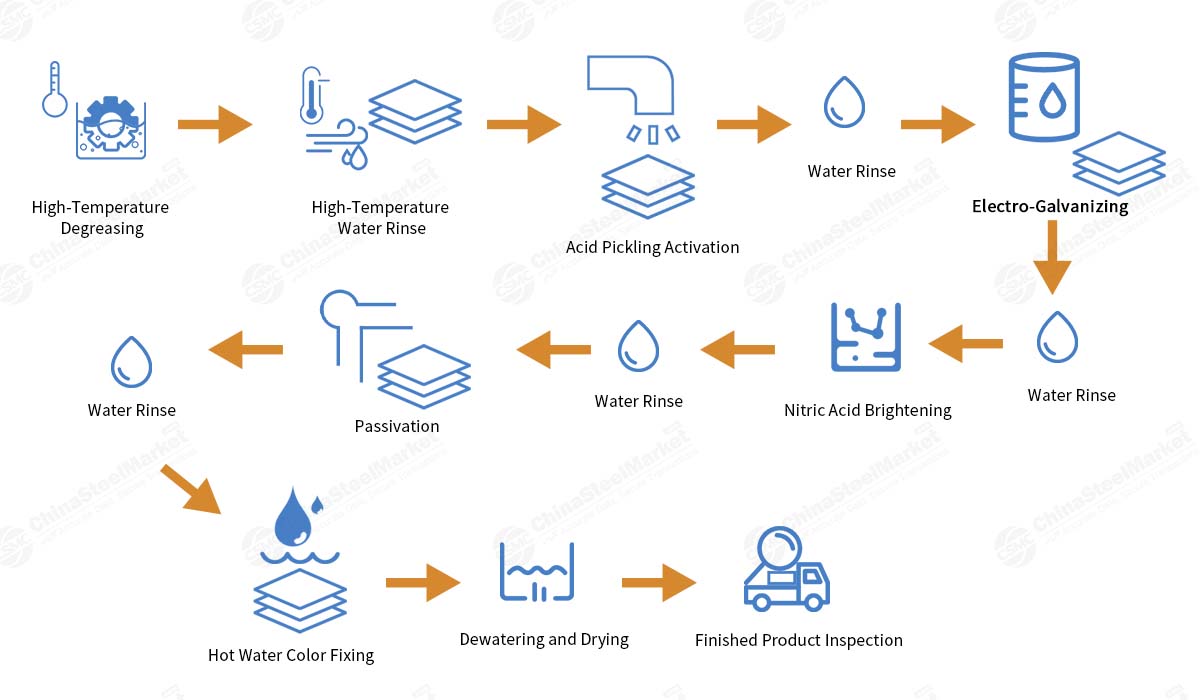
The detailed process flow is as follows:
Surface pretreatment:
Degreasing: Removing grease, stains, and impurities from the workpiece surface using chemical or electrolytic methods; alkaline degreasing solutions are typically used.
Rinsing: After degreasing, the workpiece is rinsed thoroughly with plenty of clean water to prevent residual alkaline solution from contaminating subsequent processes.
Acid Pickling: The workpiece is immersed in an acid solution (such as dilute hydrochloric acid or sulfuric acid) to remove surface oxides and rust layers, ensuring proper adhesion of the plating.
Activation: Further treatment with a weak acid or neutral salt solution removes passivation films and surface rust from the workpiece, enhancing the bond strength of the plating.
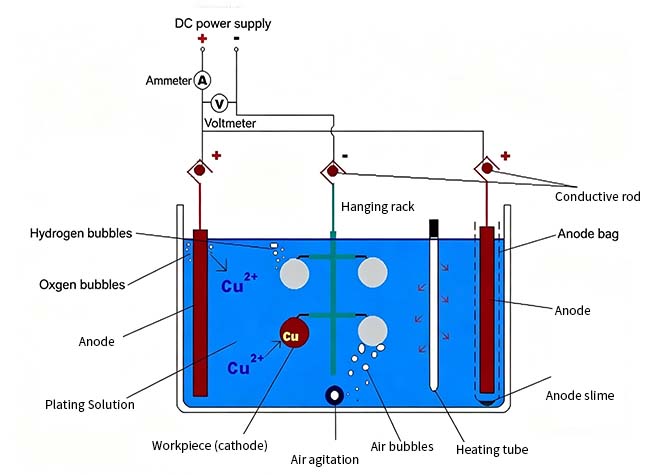
Electroplating:
Preparation of the electroplating tank: Immerse the workpiece in an electroplating solution containing zinc ions. Common electroplating baths include zinc sulfate, zinc chloride, or zinc cyanide solutions.
Current Control: Apply direct current between the workpiece (cathode) and an insoluble anode using an external power source. Under the influence of the electric field, zinc ions are reduced and deposited on the surface of the workpiece, forming a uniform coating.
Post-treatment:
Rinsing: After electroplating is complete, rinse the workpiece thoroughly with plenty of clean water to remove excess plating solution.
Passivation: To enhance the corrosion resistance of the zinc coating, the workpiece is typically immersed in a passivation solution (such as a chromate solution) after plating. The protective film formed by the passivation process significantly improves the corrosion resistance of the zinc layer.
Drying: Finally, the workpiece is dried to remove surface moisture and prevent oxidation.
In addition to hot-dip galvanizing and electrogalvanizing, for high-strength fasteners (Grade 10.9 or higher) at risk of hydrogen embrittlement, mechanical zinc plating (cold welding of zinc powder at room temperature, with no risk of hydrogen embrittlement and adjustable thickness ranging from 5 to 110 μm) or powder diffusion zinc plating (formation of an alloy layer via high-temperature diffusion, with a thickness of 30 to 80 μm and extremely strong adhesion) may also be selected. These two processes are indispensable options in specific applications.
3. The Difference Between Hot-Dip Galvanizing and Electrogalvanizing

3.1 Process Principles
Hot-dip galvanizing: Steel is immersed in molten zinc at 450–480°C, where zinc reacts metallurgically with iron to form a zinc-iron alloy layer, followed by the formation of a pure zinc layer on the surface of the alloy layer; this is a metallurgical bond.
Electrogalvanizing: Using the principle of electrolysis, steel serves as the cathode and zinc ingots as the anode. A direct current is passed through the electrolyte, causing zinc ions to reduce and deposit on the steel surface to form a zinc layer. This is a physical bonding process.
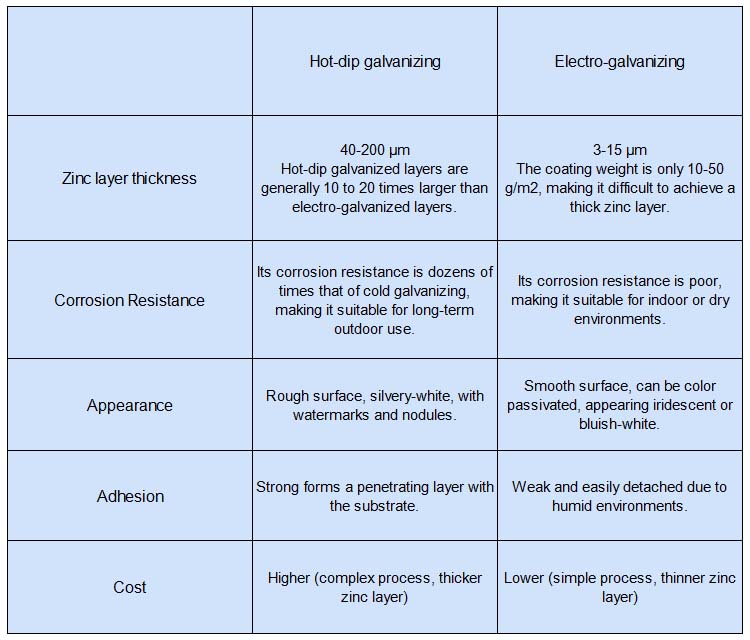
3.2 Zinc Coating Thickness
Hot-dip galvanized coatings are generally thicker, typically ranging from 40 μm to over 200 μm; in fact, they are usually 10 to 20 times thicker than electrogalvanized coatings.
In contrast, electrogalvanized coatings are very thin, typically ranging from 3 to 15 μm, with a coating weight of only 10–50 g/m², making it difficult to achieve a thick zinc layer.
3.3 Adhesion
Hot-dip galvanizing: The zinc coating bonds tightly to the steel through intermetallic compounds, resulting in strong adhesion. It is resistant to impact and abrasion, and is unlikely to peel or flake off even after long-term use.
Electrogalvanizing: The coating has a weaker bond with the substrate. Friction, impact, or prolonged exposure can easily cause the coating to peel off, resulting in poor durability.

Coating adhesion was tested using a press. The figure shows zinc-coated samples with varying levels of coating adhesion observed during the test. Samples with good coating adhesion showed no peeling of the zinc layer after pressing, whereas samples with poor adhesion exhibited coating peeling in the areas subjected to deformation.
3.4 Corrosion Resistance
Hot-dip galvanizing: Provides both physical barrier protection and electrochemical protection. If the zinc coating is damaged, the zinc acts as a sacrificial anode to continue protecting the steel substrate. It offers strong corrosion resistance and is suitable for harsh environments, with a service life of 15–50 years in outdoor or highly corrosive environments.
Electrogalvanizing: Relies solely on physical barrier protection. Once the coating is damaged, the steel substrate is directly exposed and prone to rust. It is suitable only for mildly corrosive environments; indoors, it typically lasts 3–5 years, while outdoors or in humid environments, rust may appear within about one year.
3.5 Service Life
Hot-dip galvanizing typically has a service life of 20–50 years due to its thick coating (>65 μm) and dense alloy layer, which provide exceptional corrosion resistance; in contrast, electrogalvanizing has a service life of only 3–8 years because its coating is thin (5–15 μm) and porous, resulting in poor corrosion resistance.
3.6 Appearance
Hot-dip galvanizing: The surface has a matte silver-gray finish and may exhibit zinc bloom, water marks, or drips; the overall texture is relatively rough.
Electrogalvanizing: The surface is smooth and shiny; it can be treated with colored passivation for a more aesthetically pleasing appearance.
3.7 Costs
Hot-dip galvanizing requires a significant capital investment in equipment, including the construction of dedicated zinc pots, pretreatment tanks, and other facilities. Additionally, maintenance costs for zinc pots are high due to the need for zinc dross removal and repairs to inner walls caused by corrosion.
In terms of raw materials, hot-dip galvanizing requires a large amount of zinc ingots, and fluctuations in zinc prices can have a certain impact on costs.
Regarding energy consumption, the hot-dip galvanizing process generally requires heating the molten zinc to approximately 440–465°C for 0.5–1.5 minutes, resulting in high energy consumption.
Regarding labor costs, because the hot-dip galvanizing process is relatively complex, traditional production lines require a large number of operators to perform pre-treatment, galvanizing, and post-treatment tasks, resulting in relatively high labor costs. Overall, the initial costs of hot-dip galvanizing are relatively high; however, due to its excellent corrosion resistance, it can reduce long-term maintenance costs, making it a cost-effective solution in the long run.
The main equipment for electrogalvanizing includes electroplating tanks, power supplies, and hanging fixtures. The equipment is relatively simple, and the initial investment cost is lower than that of hot-dip galvanizing.
In terms of raw materials, the cost of zinc salts and other materials used in electrogalvanizing is relatively low.
In terms of energy consumption, the electricity consumption during the electroplating process is significantly lower than the gas/electric heating energy consumption required for hot-dip galvanizing.
Regarding labor costs, manual production lines (for plating small parts) require a larger number of operators (for pretreatment, loading/unloading, and post-treatment), with labor costs accounting for approximately 20% of total costs; automated production lines (such as barrel plating lines and continuous strip plating) have lower labor costs. Therefore, the total cost of electrogalvanizing is relatively low, making it suitable for large-scale industrial production.
3.8 Applications
Hot-dip galvanizing is suitable for large structural components and parts that require high corrosion resistance and are exposed to harsh environments. In the construction industry, this includes building structures, roofs, columns, guardrails, and similar components; In the transportation sector, applications include railway and highway guardrails, bridge structural components, etc.; in the power industry, transmission towers and metal structures in substations, etc.; and in the petrochemical industry, pipelines and equipment supports, etc. Metal products in these fields are often exposed to the outdoors for extended periods or come into contact with corrosive media; hot-dip galvanizing provides reliable protection and extends their service life.
Electrogalvanizing is suitable for products that require high surface quality and dimensional accuracy, along with a certain level of corrosion resistance, but are used in relatively less harsh environments. Examples include: electronic product housings and internal components in the electronics industry; small components and trim parts in automotive manufacturing; various tools, locks, and bathroom fixtures in the hardware sector; small metal decorative elements in architectural decoration; and housings and internal components of instruments and meters.

4. International Standards for Zinc Coating Thickness
Zinc coating thickness is one of the key indicators for assessing the quality of galvanizing, and both national and international standards provide clear specifications for this parameter. These standards are generally divided into two categories: general hot-dip galvanizing standards applicable to general steel products, and specialized standards for specific products such as fasteners.
4.1 General Standards for Hot-Dip Galvanizing
The most commonly used general standards for hot-dip galvanizing include ISO 1461, ASTM A123, and China’s GB/T 13912. These standards specify minimum zinc coating thicknesses based on the thickness of the steel substrate, rather than differentiating by service environment or product application. The table below uses ISO 1461 as an example:
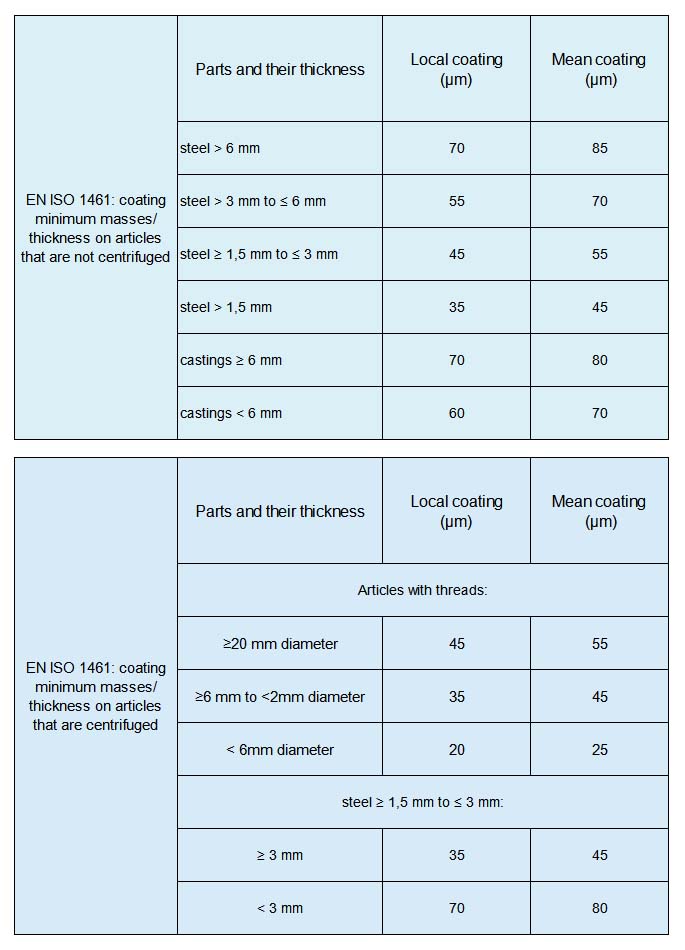
The above data represents general requirements; specific projects should refer to the latest version of the standards and the customer’s technical specifications.
Hot-dip galvanizing is primarily intended for outdoor applications requiring heavy-duty corrosion protection, such as steel components, bridges, and outdoor structural elements. The table above provides a detailed overview of the core general standard, ISO 1461, which specifies minimum zinc coating thicknesses based on the thickness of the steel substrate. The minimum coating thicknesses for the most common non-centrifugal products are as follows: when the substrate thickness is ≤1.5 mm, the minimum local thickness is 35 μm; 45 μm for base material thickness >1.5 mm and ≤3 mm; 55 μm for >3 mm and ≤6 mm; and 70 μm for >6 mm, with a minimum average thickness of 85 μm. Castings have slightly higher requirements (e.g., for castings <6 mm, the minimum local thickness is 60 μm and the minimum average thickness is 70 μm). The U.S. ASTM A123/A123M standard is also based on substrate thickness, specifying a thickness range of approximately 45 to 100 μm or more. The European standard EN ISO 1461 is essentially consistent with ISO 1461. The thickness of a hot-dip galvanized coating can also be expressed in terms of zinc coating weight (g/m²), with a conversion ratio of approximately 1 μm ≈ 7.14 g/m²; common designations include Z100, Z200, etc.
To ensure coating quality, international standards also specify adhesion test methods, such as the bend test (checking whether the coating cracks or peels off after the specimen is bent 180°) and the cross-cut test (scratching a 1 mm × 1 mm grid, with the peel area required to be ≤5%).
Hot-dip galvanized coatings are designed to prevent corrosion of steel components. The duration of the coating’s corrosion protection (whether the coating is bright or dark gray) is roughly proportional to the coating thickness. For components operating under extremely severe corrosion conditions or requiring a longer service life, the coating thickness may need to exceed the specified minimum.
When a thicker coating is required, the supplier and the customer should specify the relevant technical conditions (e.g., sandblasting treatment, chemical composition of the substrate) in the agreement.
When selecting a galvanizing standard, one should comprehensively consider environmental corrosivity (higher-humidity, coastal, or industrial environments require thicker coatings, such as ISO 1461 or ASTM A123), cost (hot-dip galvanizing costs approximately 5%–8% more for every 10 μm increase in thickness), and specific applications (hot-dip galvanizing for outdoor structures, electrogalvanizing for indoor precision components). Additionally, specific industries (such as power and oil and gas) may have additional requirements. Overall, hot-dip galvanizing is a reliable choice for heavy-duty outdoor corrosion protection, while electrogalvanizing is suitable for indoor applications with strict dimensional and aesthetic requirements.
There is no single “standard value” for galvanized coating thickness; the key lies in matching the correct standard system based on the substrate thickness of the workpiece and the application scenario. During acceptance testing, it must be clarified whether the assessment is based on local minimum thickness or average thickness.
4.2 Electrogalvanizing and Fastener Standards
In addition to general hot-dip galvanizing standards, electrogalvanizing and fastener products have their own specific thickness standard systems, as detailed below:
Electrogalvanized coatings are highly pure and feature a dense, uniform structure. They provide electrochemical protection for the steel substrate and exhibit high chemical stability at room temperature. The coating is relatively thin, generally ranging from 5 to 25 μm. Due to the low hardness of the zinc coating, it is not suitable for applications subject to friction and wear. Zinc coatings are widely used for corrosion protection and decoration on mechanical parts, wire, sheet metal, fasteners, electrical components, and instrument panels.
Representative standards for electrogalvanized products include:
a. ISO 2081: General industrial applications, with typical thicknesses of 5–25 μm; functional coatings (such as for automotive parts) may exceed 30 μm.
b. EN 10346 (continuously hot-dip galvanized steel sheets, thickness 5–20 μm),
c. High-performance ISO 19598 (suitable for high-stress automotive components, with typical values of 5, 8, and 12 μm, and requiring hexavalent chromium-free passivation),
d. ASTM B633 (based on corrosion severity, electrogalvanized coatings are classified from SC0 (no protection required) to SC4 (severe environments), with corresponding thicknesses typically ranging from 5 to 25 μm),
e. GB/T 9799: Similar to ISO 2081, this standard classifies coatings based on environmental corrosion resistance requirements and specifies different thickness ranges. For example, general environments: 5–8 μm; severe environments: ≥12 μm.
The required thickness of the zinc coating depends on the severity of the operating conditions and the expected service life; as operating conditions become more severe and the service life is extended, the thickness of the zinc coating should be increased accordingly. Depending on the specific operating environment, zinc coatings of different thicknesses can be applied, classified as SC4, SC3, SC2, and SC1, with minimum thicknesses of 25 μm, 12 μm, 8 μm, and 5 μm, respectively.
SC1—Mild—Used in indoor environments with minimal condensation and slight wear. Examples: buttons, metal wire tools, fasteners.
SC2—Moderate—Used primarily in dry indoor environments with occasional condensation and wear. Examples: tools, zippers, shelves, machine parts.
SC3 — Severe — Condensation, moisture penetration; not frequently exposed to rain or cleaning agents.
SC4 — Very Severe — Exposed to extremely harsh environments, or frequently exposed to moisture, cleaning agents, salt solutions, and the effects of impact, friction, or abrasion.
The thickness of electrogalvanized coatings is significantly lower than that of hot-dip galvanized coatings, and is expressed in grades: Fe/Zn 5 (5 μm), Fe/Zn 8 (8 μm), Fe/Zn 12 (12 μm), and Fe/Zn 25 (25 μm). In dry indoor environments, a thickness of 5–12 μm is sufficient to meet requirements, while outdoor or humid environments require 12–25 μm.
For hot-dip galvanized fasteners, ISO 10684 specifies a minimum thickness of 55 μm for thread diameters ≥ 6 mm and 45 μm for diameters less than 6 mm. It should be noted that hot-dip galvanizing may affect thread fit accuracy; therefore, sufficient tolerances should be allowed for during the design phase, or tapping should be performed after galvanizing.
In general, the thickness requirements specified in the standards represent the minimum threshold for ensuring corrosion resistance. When selecting products, these standards should serve as a basis, and the thickness should be appropriately increased in consideration of the operating environment and design life, rather than mechanically applying the minimum values.
4.3 Methods for Measuring Coating Thickness:
Coating thickness is a key indicator of coating quality. It directly affects the workpiece’s corrosion resistance and electrical conductivity, which in turn significantly impacts the product’s reliability and service life. Therefore, accurate methods for measuring coating thickness are particularly important.
4.3.1 Destructive Methods
Destructive methods are a group of techniques characterized by the destruction or damage of the coating under test during the testing process. These methods include:
Dissolution Method: This method determines the average thickness of the entire coating by immersing the specimen in a suitable solution to dissolve the coating or the base metal, and then comparing the specimen’s weight before and after dissolution or using chemical analysis to determine the coating’s mass.
Coulometric Method (Electrolytic Method): The coulometric method applies a constant direct current through an electrolytic solution to dissolve the coating metal at the anode, calculating the coating thickness based on the amount of charge consumed. This method is suitable for determining the local thickness of single-layer or multi-layer single-metal coatings.
Chronological liquid flow method: This method dissolves a localized portion of the coating on the sample under the action of a liquid flow, and the coating thickness is calculated by recording the time required for dissolution.
4.3.2 Non-destructive Methods
Non-destructive methods are a set of techniques characterized by the fact that they do not damage the coating being tested during the measurement process. These methods include:
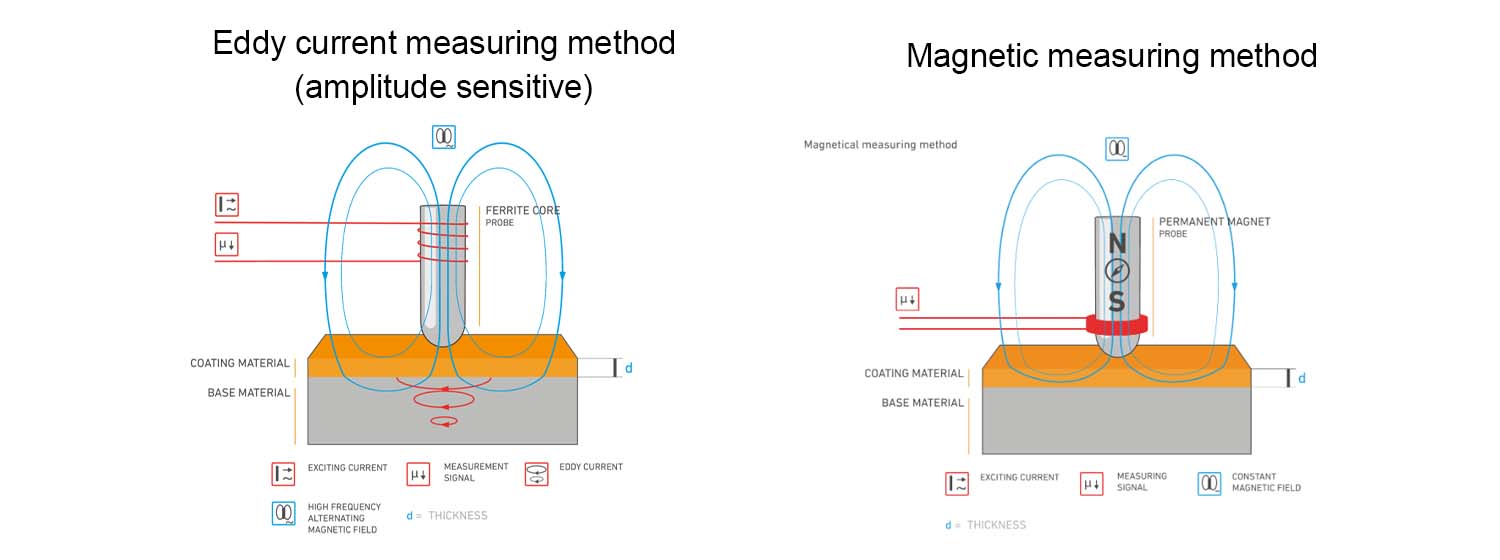
Magnetic Method: Suitable for measuring the thickness of non-magnetic coatings on magnetic metal substrates. This method typically yields small measurement errors, but its accuracy may be slightly lower for very thin coatings.
Eddy Current Method: This method uses a high-frequency coil to generate a high-frequency magnetic field, inducing eddy currents within the test specimen. The coating thickness is determined based on the amplitude and phase of these eddy currents. This method is suitable for measuring the thickness of non-conductive coatings on non-magnetic metal substrates.
β-ray reflection method: This method uses a radioactive isotope to emit X-rays, and the thickness of the coating is determined based on the intensity of the β-rays reflected by the coating. It is suitable for thin precious metal coatings.
X-ray fluorescence(XRF) method: By measuring the secondary X-rays generated at the metal surface, this method can measure the thickness of metal coatings on metal or non-metal substrates and can also determine the composition of alloy coatings.
4.3.3 Other Methods
In addition to the destructive and non-destructive methods described above, there are several other techniques used to measure coating thickness, such as metallographic microscopy, profilometry, interferometric microscopy, and dual-beam microscopy. Each of these methods has its own advantages and is suitable for specific applications; the appropriate method can be selected for testing as needed.
5. Key Factors Affecting the Selection of Zinc Coating Thickness: Selection Guidelines and Common Misconceptions
When selecting the appropriate zinc coating thickness, it is necessary to comprehensively evaluate the operating environment, substrate properties, workpiece structure, design life, and process compatibility to avoid one-sided decisions based on the notions that “thicker is better” or “thinner is more cost-effective.”
5.1 Key considerations in selecting solutions for actual projects.
5.1.1 Corrosion Class of the Operating Environment
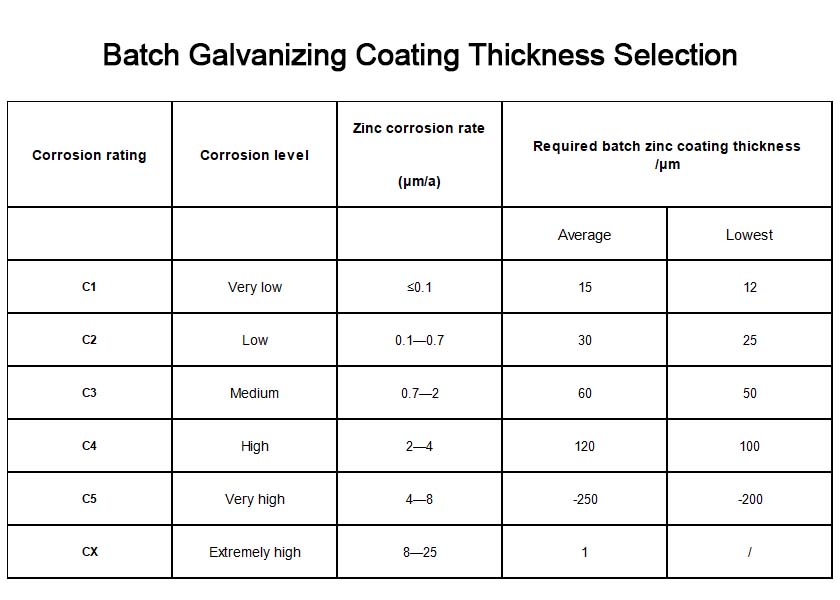
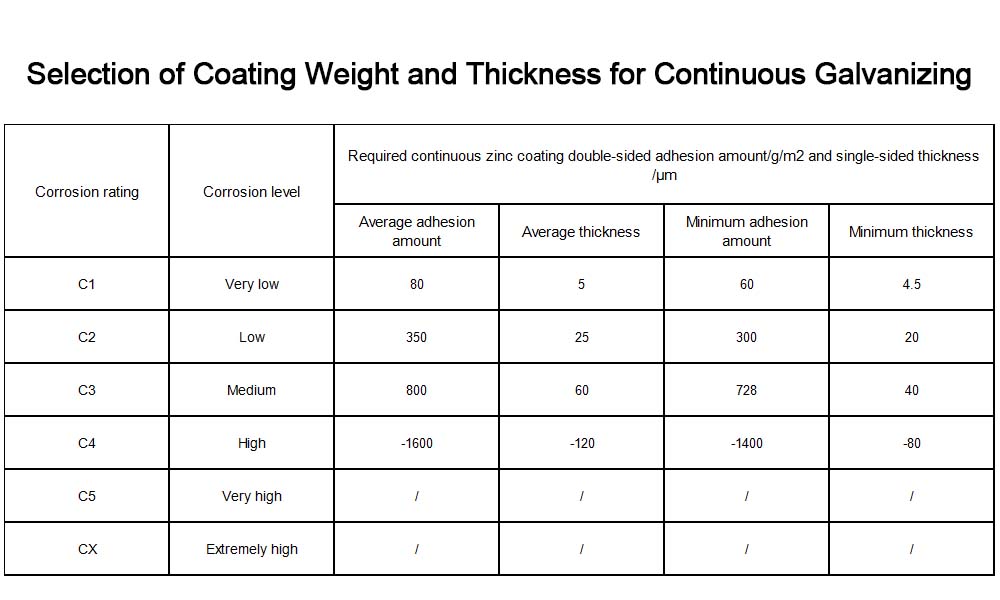
The corrosion class of the operating environment is the primary factor in product selection. ISO 9223 classifies atmospheric corrosion into C1 through C5 and CX classes.
C1: Very low corrosion: e.g., dry indoor environments such as offices and museums. Average zinc coating thickness ≥ 65 μm, local thickness ≥ 55 μm. Electrogalvanizing is sufficient, and the thickness may be appropriately reduced.
C2 (Low Corrosivity): Temperate rural areas and low-pollution atmospheric environments. Average zinc coating thickness ≥80 μm, local thickness ≥65 μm. Electrogalvanizing is sufficient to meet requirements.
C3 (Moderate Corrosivity): Urban outdoor environments and general industrial facilities. Recommended hot-dip galvanizing thickness ≥65 μm. The average annual corrosion rate of the zinc coating in this environment is approximately 0.85 μm.
C4 (High Corrosion): Chemical plants, coastal industrial zones. Hot-dip galvanizing thickness is recommended to be >65 μm, or zinc-aluminum-magnesium coatings may be considered.
C5–CX (Very High to Extreme Corrosion): High-salinity coastal and offshore environments. Hot-dip galvanizing thickness must reach 140 μm or more.

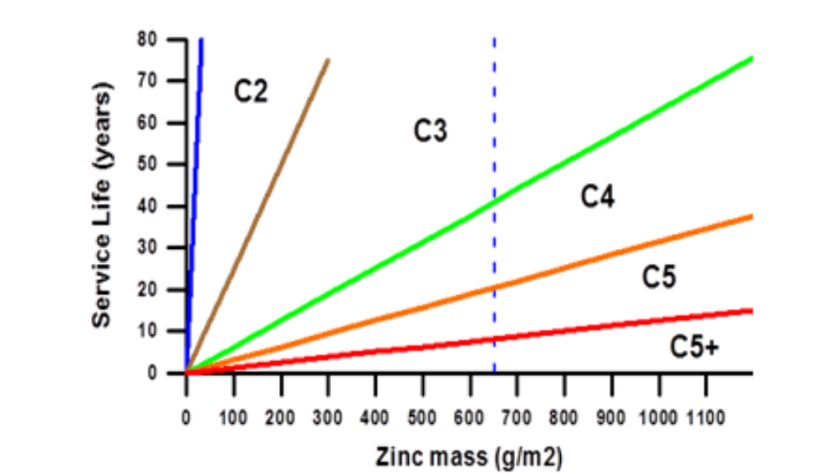
Service Life of Galvanized Coatings by Coating Weight and Corrosive Environment
Key Selection Criteria: Determine the coating thickness based on the specific corrosion class of the project; indoor standards must not be applied to outdoor applications. Additionally, the corrosion levels from C1 to C5 do not follow an arithmetic progression but rather a geometric progression, with each level representing approximately a twofold increase. The following two tables show the required batch galvanized coating thicknesses, as well as the continuous galvanized coating adhesion and thickness, for different corrosion environment classes when the service life is 25 years.
Due to process limitations, the actual coating thickness has a minimum and maximum limit. In actual production, C1 and C2 grades are generally controlled at the minimum thickness that can be produced. The thickness required for C5 grade is very difficult to produce, while CX grade cannot be met by galvanizing alone. Generally, both C5 and CX grades employ a combination of galvanizing and other composite corrosion protection measures.
5.1.2 Base Material Composition of Steel
Due to the vigorous reaction between iron and zinc, a high carbon content in steel may result in a thinner coating. Additionally, the silicon (Si) and phosphorus (P) content in steel can affect the thickness and microstructure of the hot-dip galvanized coating. When the silicon content exceeds 0.07%, abnormal thickening of the coating may occur (the St. Dering effect). This effect is particularly pronounced in active steels with silicon (Si) content ranging from 0.12% to 0.25%.
When selecting materials, it is important to understand the silicon content of the steel. For reactive steels, the zinc bath can be adjusted by adding elements such as nickel, or by modifying the galvanizing temperature and duration, to achieve the desired coating thickness and adhesion.
A high silicon content in the steel substrate can pose challenges for hot-dip galvanizing. It is generally accepted that killed steel with a high silicon content is unsuitable for hot-dip galvanizing; therefore, for many years, boiling steel with a silicon content of less than 0.07% has been the primary choice for hot-dip galvanizing base steel.
Experience has shown that a high silicon content in the steel substrate causes a significant thickening of the ξ-phase iron-zinc alloy layer in the galvanized coating, resulting in a gray coating and poor adhesion of the galvanized layer.
It is widely recognized that high silicon content in steel substrates has an adverse effect on hot-dip galvanizing. However, the underlying mechanism is quite complex; generally, it is believed that the impact of silicon in steel sheets on hot-dip galvanizing depends primarily on silicon dioxide rather than on free silicon.
In addition, the presence of phosphorus as an impurity in steel can cause cold brittleness. Therefore, phosphorus content is typically kept very low during production, generally below 0.05%. Steel with this composition does not cause significant issues during galvanizing. Even high-phosphorus thin sheets with phosphorus content ranging from 0.06% to 0.12% do not exhibit any noticeable abnormalities during hot-dip galvanizing. However, when the phosphorus content in steel reaches 0.15%, the galvanized coating forms an exceptionally thick Fe-Zn alloy layer that is prone to cracking. The η phase becomes thinner, while the ζ and δ1 phases grow more rapidly. This indicates rapid diffusion of iron atoms, causing the surface of the galvanized sheet to become dull and lose its pattern, forming spots, while the adhesion of the coating also deteriorates.
5.1.3 Steel Structure and Processing Technology
Effect of wall thickness: Thin-walled steel dissipates heat quickly, making it difficult to form a thick coating. Standards (such as ISO 1461) specify reference coating thicknesses based on the substrate thickness.

Complex Shapes: In areas such as grooves, corners, and enclosed cavities, the flow of the plating solution is restricted, which may result in localized coating thickness failing to meet specifications. Appropriate design allowances should be added, or a composite coating should be used.
Processing Performance: Thicker coatings are more prone to cracking and peeling during bending and stamping. The appropriate thickness should be selected based on whether subsequent forming processes are required.
Process Parameters: The temperature (440–465°C) and immersion time for hot-dip galvanizing directly affect the coating thickness; the current density for electrogalvanizing must be precisely controlled.
5.2 Selection criteria and engineering application recommendations
Choosing the appropriate zinc coating thickness is a complex undertaking that requires a comprehensive balance between cost and corrosion protection lifespan. The key lies in accurately matching the application environment with the expected corrosion protection lifespan and finding a balance between cost and process coordination. Depending on the specific process and environment, the final coating thickness is a crucial physical indicator determining the corrosion protection lifespan.
Environmental Determination Method: The first step is to determine the atmospheric corrosion rating (C1-CX) of the product's intended use environment, and then select the minimum recommended thickness according to the standard.
After determining the environmental rating, calculate the required thickness based on the expected maintenance-free years and the corrosion rate of the galvanized layer (e.g., in a typical atmospheric environment, the corrosion rate of hot-dip galvanizing is approximately 1-2 micrometers/year).
Here are specific thickness recommendations based on application scenarios:
1. Outdoor heavy-duty corrosion protection: Hot-dip galvanizing is preferred, with a thickness ≥70μm, and zinc layer loss should be checked regularly.
2. Precision assemblies: Electro-galvanizing (8-12μm) is recommended to avoid excessively thick coatings affecting tolerances.
3. Indoor environment: For indoor components with less stringent corrosion protection requirements, electro-galvanizing (5-10μm) or continuous hot-dip galvanized thin sheet (5-20μm) can be used. If batch hot-dip galvanizing is used, the thickness is usually not less than 35μm.
4. General environment: For general outdoor environments (corresponding to C2-C3): A hot-dip galvanized layer thickness ≥45-60μm is recommended, with a service life of 10-20 years.
5. Severely Corrosive Environments (Corresponding to C4): In severely corrosive environments, such as coastal areas and chemical plants, the zinc layer thickness of hot-dip galvanizing needs to be further increased, generally to 70-100 μm. This thickness of zinc layer can protect the metal substrate for a long time in harsh environments, with a service life of 20-50 years.
6. C5 High Corrosion Level (Corresponding to C5-CX): The zinc layer thickness of C5 anti-corrosion hot-dip galvanizing is typically required to be between 80-120 μm, far exceeding the requirements for general environments. It can maintain the surface of metal products intact for a long time in extreme environments.
In practical engineering, there are many misconceptions regarding the selection of zinc coating thickness. Some believe that the thicker the zinc coating, the more reliable and effective the corrosion protection. However, excessively thick coatings (such as hot-dip galvanizing exceeding 120 micrometers) become significantly more brittle, easily crumbling and peeling off under impact or bending, and costs increase dramatically. Therefore, the thickness should be appropriate.
Another common misconception is that as long as the average thickness meets the standard, slightly thinner areas are acceptable. In fact, corrosion often begins at the weakest point of the coating; excessively thin areas lead to premature rusting. Therefore, standards specifically emphasize that the minimum thickness in localized areas must meet the specified value.
Some users mistakenly believe that electroplating can replace hot-dip galvanizing for long-life outdoor applications. In reality, the maximum thickness for electroplating is only about 25 micrometers, and the corrosion resistance of pure zinc is weaker than that of the iron-zinc alloy layer in hot-dip galvanizing. Therefore, hot-dip galvanizing or mechanical galvanizing should still be used in harsh outdoor environments.
With increasingly stringent environmental regulations and advancements in materials science, the selection of zinc coating thickness is shifting from "experience-driven" to "data-driven". The selection of zinc coating is not about pursuing the ultimate thickness, but about pursuing the right match. It is a systematic project that takes the application environment and expected life as its starting point, and ultimately locks in a thickness solution that is both technically and economically sound by selecting a matching galvanizing process.
We sincerely hope that the information we provide can make more beneficial value. In addition, we sincerely invite you to leave valuable comments and advice on our website. We will follow up on your comments and advice at any time on our website.
CSMC - Empowering small and medium-scale steel purchasing.
Editor: Hana Kyra
Mail: cs@chinasteelmarket.com



|

|

|

|

|
| Timely Info | Independent | Platform | Multiple guarantees | Self-operated storage |
| About us | Channel | Useful tools |
|---|---|---|
| About China Steel Market | Prices | Steel Weight Calculation |
| Contact Us | Answers | Why Choose Us |
| Terms & Conditions | Inventory | |
| Privacy Policy | Help |
Hot search words: